Which factor causes the slight deviation of bond angles from the idealized VSEPR angles in real molecules like chloromethane?
Answer
The electronegativity of the surrounding atoms
While the primary geometry is determined by domain count, differences in the surrounding atoms' electronegativity alter the electron density distribution, subtly changing the effective size and repulsion of specific bonds, thus slightly adjusting the bond angles.
Related Questions
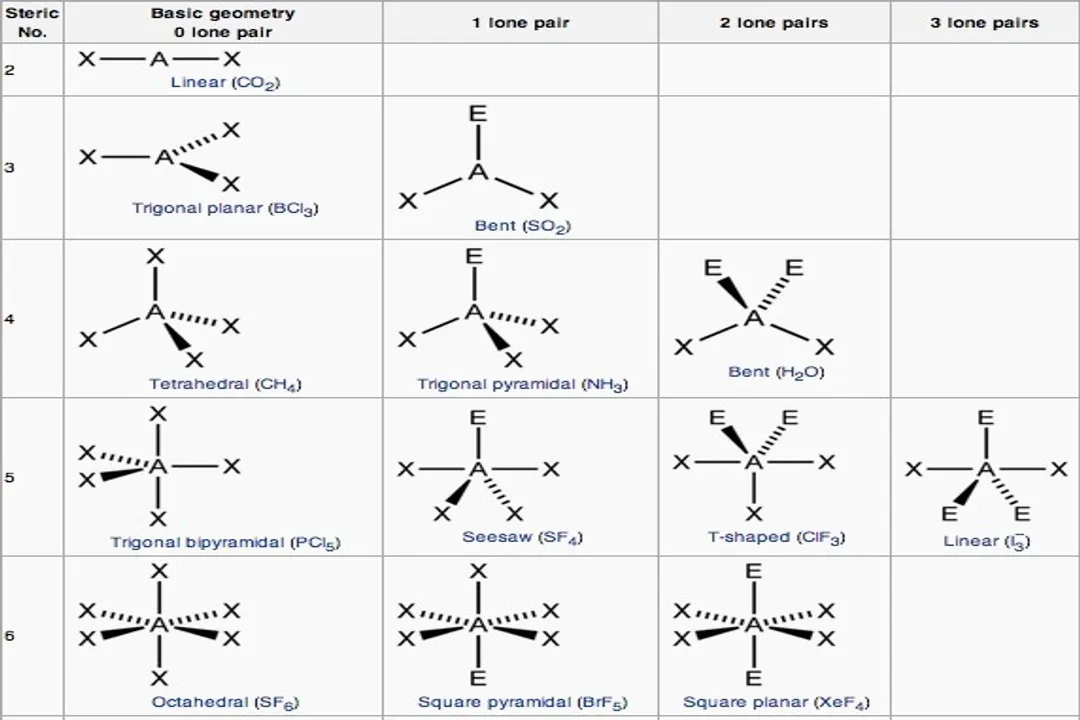
What constitutes an electron domain in the VSEPR model?For the purpose of determining initial geometry, how is a triple bond counted regarding electron domains?What is the order of repulsion strength between electron domains, from greatest to least?Why do lone pairs exert stronger repulsion than bonding pairs?What molecular geometry results when a molecule has a tetrahedral electron geometry with two bonding pairs and two lone pairs ($ ext{AX}_2 ext{E}_2$)?What is the electron geometry when a molecule has five total electron domains?What molecular geometry is characteristic of a molecule with five domains where one domain is a lone pair ($ ext{AX}_4 ext{E}$)?In an Octahedral electron geometry (6 domains), what shape is formed if there are two lone pairs ($ ext{AX}_4 ext{E}_2$)?What is the approximate bond angle found in water ($ ext{H}_2 ext{O}$), which has two lone pairs on the central oxygen atom?Which factor causes the slight deviation of bond angles from the idealized VSEPR angles in real molecules like chloromethane?