bond articles
What property describes a mineral's resistance to scratching or permanent deformation?

What is the most significant factor determining a mineral's hardness?

Which bond type results in exceptionally tough and hard structures like Diamond?

What typically causes an ionic crystal to cleave or break relatively easily when scratched?

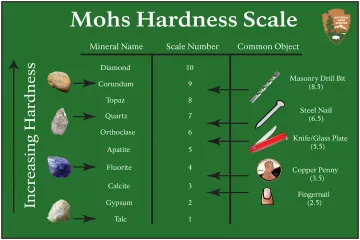
Who developed the relative hardness scale commonly used by geologists today?

How is the Mohs scale accurately described regarding measurement type?

How does Graphite differ structurally from Diamond, resulting in a large difference in hardness?

What term describes hardness varying based on the direction the scratch test is applied?

What common object approximates a hardness of about 5.5 in field testing according to the provided chart?

Which mechanical property, independent of hardness, describes a mineral's resistance to breaking or crushing?

What does electronegativity measure?

What is the maximum value assigned to an element on the Pauling electronegativity scale?

How does electronegativity generally change when moving down a column (group) on the periodic table?

What condition creates bond polarity?

What $\Delta \text{EN}$ range typically characterizes a polar covalent bond?

Why is Carbon Dioxide (CO2) considered a nonpolar molecule despite having highly polar C=O bonds?

What factor is crucial in determining the overall polarity of a molecule, even if its individual bonds are polar?

Which element is cited as having the lowest electronegativity value on the Pauling scale, making it prone to losing electrons?

How does electronegativity differ fundamentally from electron affinity or ionization energy?

Besides the electronegativity difference, what physical factor modulates the final measurable dipole moment of a bond?
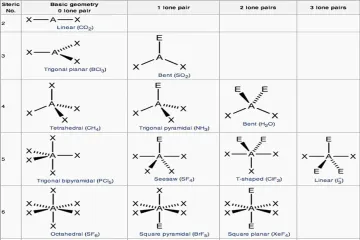
What constitutes an electron domain in the VSEPR model?

For the purpose of determining initial geometry, how is a triple bond counted regarding electron domains?

What is the order of repulsion strength between electron domains, from greatest to least?

Why do lone pairs exert stronger repulsion than bonding pairs?

What molecular geometry results when a molecule has a tetrahedral electron geometry with two bonding pairs and two lone pairs ($ ext{AX}_2 ext{E}_2$)?

What is the electron geometry when a molecule has five total electron domains?

What molecular geometry is characteristic of a molecule with five domains where one domain is a lone pair ($ ext{AX}_4 ext{E}$)?

In an Octahedral electron geometry (6 domains), what shape is formed if there are two lone pairs ($ ext{AX}_4 ext{E}_2$)?

What is the approximate bond angle found in water ($ ext{H}_2 ext{O}$), which has two lone pairs on the central oxygen atom?
