How does pH affect solubility?
The way a solid dissolves in water is not always a fixed property; it can be dramatically altered by the acidity or alkalinity of the surrounding liquid. This influence of on solubility is a cornerstone concept in solution chemistry, explaining everything from how antacids work in the stomach to why certain minerals precipitate out of natural water sources. At its heart, this phenomenon is governed by acid-base reactions that couple with the dissolution equilibrium of the salt itself. [1][5][7]
# Chemical Basis
Solubility product constants, or values, define the equilibrium between a sparingly soluble ionic solid and its constituent ions in a saturated solution at a given temperature. [1][5] For a generic salt , the equilibrium is described by . [5] When the of the solution changes, the concentration of either or ions changes, and if one of the ions released from the solid— or —reacts with these excess or , the system must shift to re-establish the balance. [6] This shift, predicted by Le Chatelier’s principle, means the solid must dissolve further to replenish the consumed ion, thereby increasing the overall solubility. [1][6]
For to have a significant effect, at least one of the ions produced upon dissolution must be capable of acting as an acid or a base in water. [1][7] If a salt is composed of ions derived from a strong acid and a strong base (like ), neither ion will significantly react with added or , and thus the solubility is effectively independent of . [5][7]
# Acid Salts
When a salt contains an anion that is the conjugate base of a weak acid, lowering the (making the solution more acidic) will cause the anion to become protonated. [1][5] This effectively removes the anion from the solution equilibrium, forcing more of the solid salt to dissolve. [6]
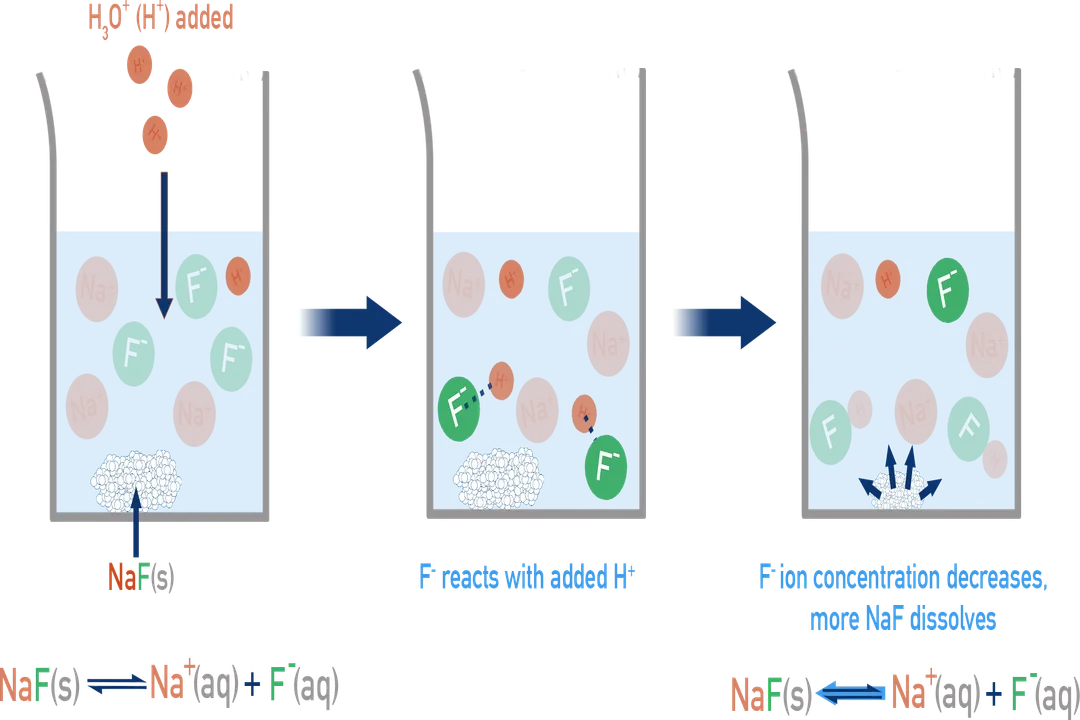
Consider the example of calcium fluoride, , which has a small of . [1] The dissolution equilibrium is:
The fluoride ion, , is the conjugate base of the weak acid hydrofluoric acid (). When acid () is added, it reacts with the fluoride ions:
Because the fluoride ions are being consumed in this secondary reaction, the equilibrium for the dissolution of shifts to the right, increasing the concentration of and, overall, dissolving much more solid than would occur in a neutral solution. [5][7] This reaction is a classic illustration of the common ion effect in reverse—the addition of acts to remove one of the product ions. [1]
A similar mechanism occurs with salts derived from weak bases. If the cation of the salt is itself the conjugate acid of a weak base, it can react with added hydroxide ions () when the solution becomes basic (). [5][7] For instance, ammonium chloride, , dissolves into and . The ammonium ion, , is a weak acid. In a basic solution, the hydroxide ions can strip a proton from the ammonium ion:
This reaction removes from the dissolution equilibrium, causing more to dissolve to maintain . [5]
The extent of this influence is directly related to the strength of the conjugate pair. A weaker acid (meaning a stronger conjugate base) will react more completely with or , leading to a greater increase in solubility when the is adjusted appropriately. [7]
# Basic Salts
Salts containing anions derived from very weak acids—meaning the anions are relatively strong bases—show the most dramatic dependency. [5] Examples include metal hydroxides and salts of weak oxyanions.
Magnesium hydroxide, , is a classic sparingly soluble base, often used as an antacid or in wastewater treatment. [1]
In acidic conditions (), the excess ions readily react with the hydroxide ions:
This rapid removal of from the solubility equilibrium forces a large amount of to dissolve. In fact, the solubility of metal hydroxides in acidic solutions is often very high because the neutralization reaction is essentially irreversible compared to typical equilibrium shifts. [1]
We can quantify this difference in behavior by comparing two hypothetical salts, (anion is conjugate of a strong acid, e.g., ) and (anion is conjugate of a weak acid, e.g., ), both with the same value:
| Condition | Salt A () Solubility | Salt B () Solubility | Governing Reaction |
|---|---|---|---|
| Neutral | Low (governed by ) | Low (governed by ) | Simple Dissolution |
| Acidic | Unaffected | Significantly Increased |
This comparison highlights that while both salts might precipitate at the same concentration in pure water, will persist far longer or dissolve much more readily when exposed to mild acid compared to . [5]
# Amphoteric Behavior
A particularly interesting subset of sparingly soluble compounds involves metal hydroxides that exhibit amphoterism. [5] Amphoteric substances can act as either an acid or a base, meaning their solubility increases in both highly acidic and highly basic solutions, creating a U-shaped solubility curve when solubility is plotted against . [1]
Common examples include the hydroxides of aluminum (), chromium (), zinc (), and lead (). [5]
Low (Acidic): Like any basic salt, the hydroxide ion () is neutralized by excess ions, causing the metal hydroxide to dissolve:
The solubility increases as drops. [1][5]High (Basic): In strongly basic solutions, the metal ion itself acts as a Lewis acid and reacts with the excess hydroxide ions to form a soluble complex ion, often a tetrahydroxo- species. [5] For aluminum:
This reaction consumes the solid , increasing its solubility again as the rises. [1]
The minimum solubility for these amphoteric hydroxides occurs at an intermediate , often near neutral, where the concentration of both (needed for dissolution by neutralization) and excess (needed for complexation) is low. [1] This concept is extremely important in environmental chemistry, as it dictates when these metals will precipitate out of solution during natural weathering or industrial effluent neutralization processes. [5]
# Practical Application Scenarios
Understanding this dependence translates directly into real-world control over chemical processes. In analytical chemistry, adjustment is routinely used to separate different metal ions based on their differing solubility products and acid/base strengths. [7] For example, if you have a mixture of metal ions, you can gradually lower the . A metal hydroxide that precipitates at a high (like ) will redissolve quickly as the drops, while a metal hydroxide whose anion is a very weak base or whose cation is not amphoteric might remain dissolved longer or precipitate at a much lower . [5]
In contexts like mineral digestion or pharmaceutical formulation, the acidity of the environment is a key factor in determining bioavailability. A drug containing a weakly acidic component might dissolve poorly in the stomach ( 1-3) if the required counter-ion is not present, but dissolve much more readily once it reaches the slightly basic environment of the small intestine. [7] Conversely, a substance that must remain solid until it reaches a specific physiological location relies on the local keeping it insoluble.
When dealing with sparingly soluble fluorides in industrial settings, such as in cooling tower water treatment or mining runoff, controlling the input of strong acids or bases is not just about preventing scale or corrosion; it is the primary mechanism for controlling the migration of the fluoride ion itself. [1] If you need to remove fluoride from water, you might raise the to precipitate it as , but if you are concerned about leaching from a geological source, knowing that an acidic seep will dramatically increase the dissolution rate of any fluoride-containing rock is critical for modeling plume movement.
If you are designing a process to selectively precipitate a substance, it is important to remember that the concentration of the reactive species ( or ) must overcome the innate of the primary salt. For instance, solubility is very sensitive to because is a strong base, meaning its neutralization by is highly favorable. However, for a salt like , the reaction is governed by the of ( for the first dissociation, though is a moderate base, ). [1] This means that while the effect exists, you might need a much lower (stronger acid) to achieve the same magnitude of solubility increase for that you see for a salt like (where the anion, , is a much stronger base). [5] The relationship isn't linear; it's tied to the acid/base dissociation constants of the respective conjugate pairs.
# Solubility Dynamics
The effect introduces a dynamic element to what is often treated as a static equilibrium constant (). While the remains constant for a specific temperature, the effective solubility—the total amount of solute that can be dissolved—changes based on the solution's . [5] This is sometimes referred to as conditional solubility.
In summary, the rule of thumb is straightforward: affects the solubility of any salt that contains an ion capable of acting as a Brønsted-Lowry acid or base. [7]
- Lowering (Adding Acid): Increases the solubility of salts whose anions are conjugate bases of weak acids (e.g., , , ). [1][5]
- Raising (Adding Base): Increases the solubility of salts whose cations are conjugate acids of weak bases (e.g., ) or hydroxides that are amphoteric (e.g., ). [5]
Salts formed from strong acids and strong bases, like potassium chloride () or sodium nitrate (), do not exhibit this dependency because neither nor reacts meaningfully with added or . [7] Their solubility is essentially constant across the standard range. [5]
The measurement of itself is fundamentally about the concentration of , making it an intuitive control knob for chemical equilibria involving proton transfer. Mastering this interplay allows chemists and engineers to predict, control, and exploit solubility differences, turning what might seem like a simple dissolution process into a finely tuned chemical separation technique. [1][6]
Related Questions
#Citations
16.4: The Effects of pH on Solubility
The Effect of pH on Solubility
Does anyone know how pH affects solubility?? : r/Mcat
Does pH affect solubility?
17.5: Solubility and pH
8.11 pH and Solubility - AP Chemistry
8.11 pH and Solubility | AP Chemistry
17.6 pH Effects on Solubility

