acid articles
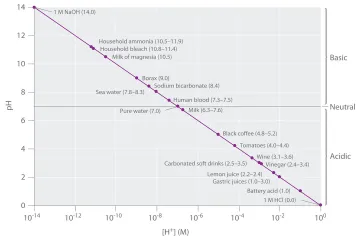
At its most basic level, what does an acid-base reaction describe?

What limitation of the Arrhenius theory is highlighted by the behavior of ammonia ($ ext{NH}_3$)?

According to the Brønsted-Lowry theory, how is an acid defined?

In the reaction $\text{CH}_3\text{COOH} + \text{H}_2\text{O} \rightleftharpoons \text{CH}_3\text{COO}^- + \text{H}_3\text{O}^+$, what does the acetate ion ($ ext{CH}_3\text{COO}^-$) represent?

What process involving water allows it to act as both an acid and a base in pure water?

What is the defining molecular event in a Lewis acid-base reaction?

Which species is defined as an electron-pair donor in the Lewis theory?

What is the relationship between Brønsted-Lowry acids and Lewis acids?

When writing the net ionic equation for aqueous neutralization, what are spectator ions?

How is the strength of an acid related to the stability of its conjugate base?
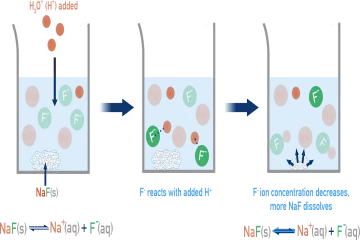
What general condition must be met for the pH of a solution to significantly alter a salt's solubility?

How is the solubility of sodium chloride ($ ext{NaCl}$) affected by changes in the solution's $ ext{pH}$?

When acid ($ ext{H}^+$) is added to calcium fluoride ($ ext{CaF}_2$), what primarily happens to the fluoride ions ($ ext{F}^-$)?

For a salt containing an anion that is the conjugate base of a weak acid, what drives increased dissolution upon lowering the $ ext{pH}$?

In a basic solution, how does the solubility of ammonium chloride ($ ext{NH}_4 ext{Cl}$) increase?

What characteristic describes the solubility curve of metal hydroxides that exhibit amphoterism when solubility is plotted against $ ext{pH}$?

Which metal hydroxides are explicitly named as common examples of amphoteric substances?

At low $ ext{pH}$, how does the dissolution of an amphoteric hydroxide like $ ext{Al}( ext{OH})_3$ proceed?

What is the mechanism causing increased solubility of amphoteric hydroxides in highly basic solutions (high $ ext{pH}$)?

If $ ext{Salt A}$ ($ ext{MCl}$) and $ ext{Salt B}$ ($ ext{MF}$) share the same $K_{sp}$, how does their solubility compare in an acidic $ ext{pH}$ environment?

What is the fundamental characteristic regarding the storage or bottling of carbonic acid?

What two substances react to fundamentally form carbonic acid?

Why does carbonic acid cause a milder effect on $ ext{pH}$ compared to hydrochloric acid?

The familiar fizz sensation in carbonated beverages is directly due to the presence of which substance?

What happens to the concentration of carbonic acid in soda when the bottle is chilled?

What narrow $ ext{pH}$ range must human blood typically maintain for essential bodily functions?

How does the respiratory system influence blood acidity related to carbonic acid regulation?

Besides manufactured drinks, where is carbonic acid naturally present?

What geological features are formed over vast timescales due to carbonic acid interaction with minerals like calcium carbonate?
