What property describes the relative ability of atoms to attract shared electrons in a chemical bond?
Answer
Electronegativity
Electronegativity is the property that defines how strongly an atom pulls shared electrons towards itself when forming a chemical bond.
#Videos
How Does Polarity Affect Solvent Choice In Chemistry? - YouTube
Related Questions
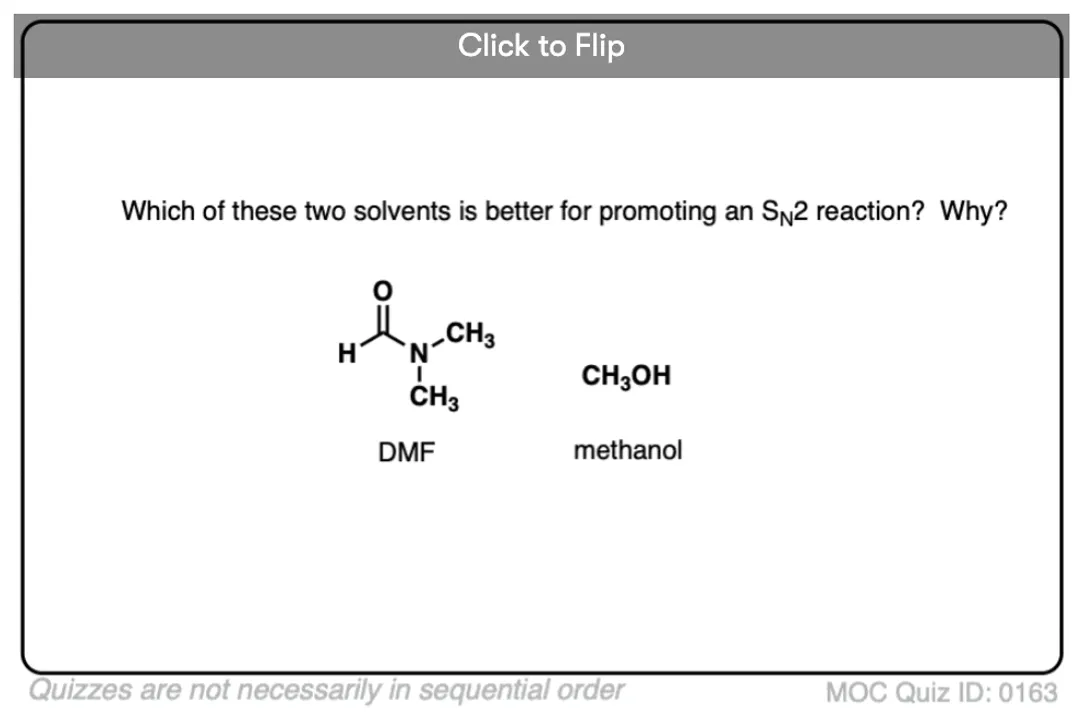
What primary property dictates compatibility and governs dissolution?What results from the uneven distribution of electrons due to differing electronegativities between bonded atoms?What property describes the relative ability of atoms to attract shared electrons in a chemical bond?What is the foundational and concise rule governing solubility?What is the process called when water forms a protective cage around ions to pull them away from a crystal lattice?Why does table salt ($ ext{NaCl}$) remain a solid crystal when placed in a nonpolar solvent like vegetable oil?What key structural feature allows Polar Protic Solvents like water to form hydrogen bonds?Compared to protic solvents, how do Polar Aprotic solvents interact less strongly with anions?What type of substances are nonpolar solvents, such as hexane and toluene, best suited for dissolving?What characteristic gives ethanol its versatile, ambivalent behavior in dissolving different types of substances?For dissolution to be energetically favorable, what compatibility factor must be high between the solute and solvent?