polarity articles

What does electronegativity measure?

What is the maximum value assigned to an element on the Pauling electronegativity scale?

How does electronegativity generally change when moving down a column (group) on the periodic table?

What condition creates bond polarity?

What $\Delta \text{EN}$ range typically characterizes a polar covalent bond?

Why is Carbon Dioxide (CO2) considered a nonpolar molecule despite having highly polar C=O bonds?

What factor is crucial in determining the overall polarity of a molecule, even if its individual bonds are polar?

Which element is cited as having the lowest electronegativity value on the Pauling scale, making it prone to losing electrons?

How does electronegativity differ fundamentally from electron affinity or ionization energy?

Besides the electronegativity difference, what physical factor modulates the final measurable dipole moment of a bond?
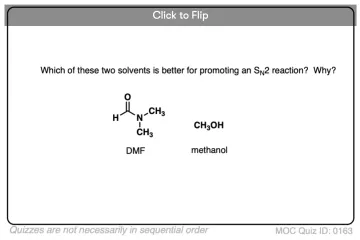
What primary property dictates compatibility and governs dissolution?

What results from the uneven distribution of electrons due to differing electronegativities between bonded atoms?

What property describes the relative ability of atoms to attract shared electrons in a chemical bond?

What is the foundational and concise rule governing solubility?

What is the process called when water forms a protective cage around ions to pull them away from a crystal lattice?

Why does table salt ($ ext{NaCl}$) remain a solid crystal when placed in a nonpolar solvent like vegetable oil?

What key structural feature allows Polar Protic Solvents like water to form hydrogen bonds?

Compared to protic solvents, how do Polar Aprotic solvents interact less strongly with anions?

What type of substances are nonpolar solvents, such as hexane and toluene, best suited for dissolving?

What characteristic gives ethanol its versatile, ambivalent behavior in dissolving different types of substances?
