What are the volatile elements on Mercury?
The innermost planet, Mercury, presents a fundamental puzzle to planetary scientists. Its searing proximity to the Sun suggests an environment utterly hostile to lighter, more easily vaporized materials, yet data from orbital missions reveals a more complex chemical inventory than that initial environmental logic would suggest. [6][8] The expectation, based on solar system formation models, was that Mercury would be dominated by high-temperature, refractory materials—elements that only condense at extremely high temperatures—while volatile elements, those that vaporize at relatively lower temperatures, would have been almost entirely stripped away during the planet's accretion. [3][8] Understanding the specific volatile elements present, and perhaps more importantly, those that are missing or surprisingly present, is key to decoding Mercury's chaotic and extreme formation history. [2][6]
# Extreme Environment
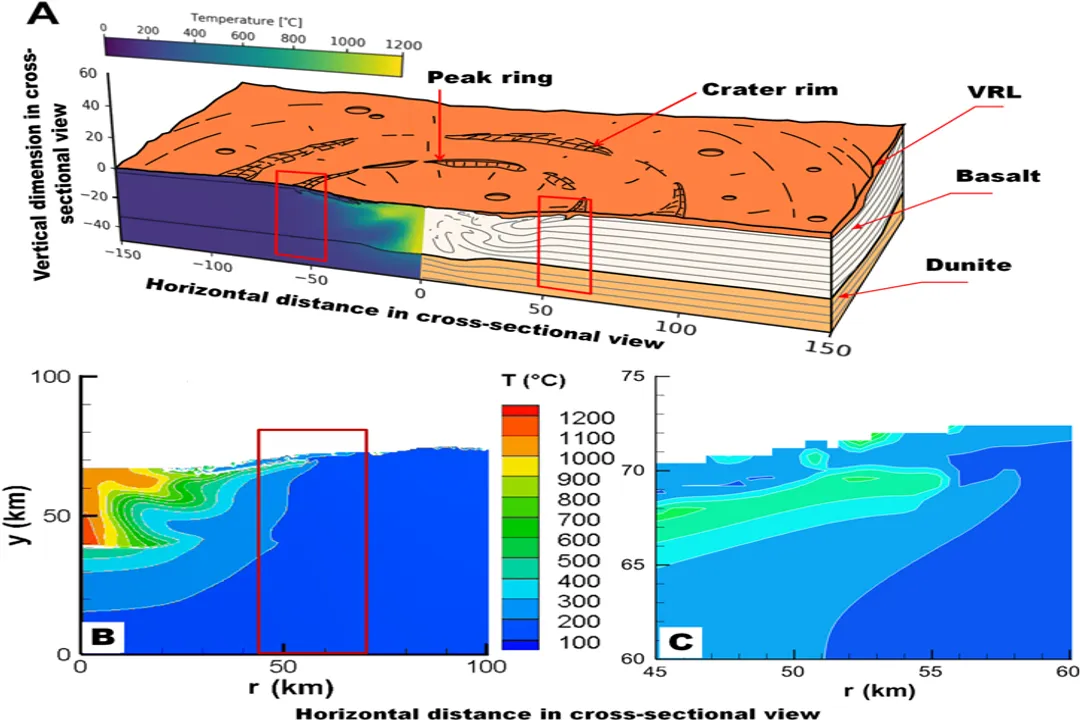
Mercury exists in an environment of intense solar radiation and extreme thermal gradients. [6] Temperatures on the sunlit side can soar, while the dark side plummets to frigid lows. [7] This proximity to the star means that any materials with lower boiling points should have been swept away by the solar wind and radiation environment during the solar system's early, dynamic history. [3] Therefore, finding any significant concentration of volatiles, even in unexpected reservoirs like the polar regions, fundamentally challenges the simplest accretion models for a terrestrial planet this close to the Sun. [1][6]
# Compositional Evidence
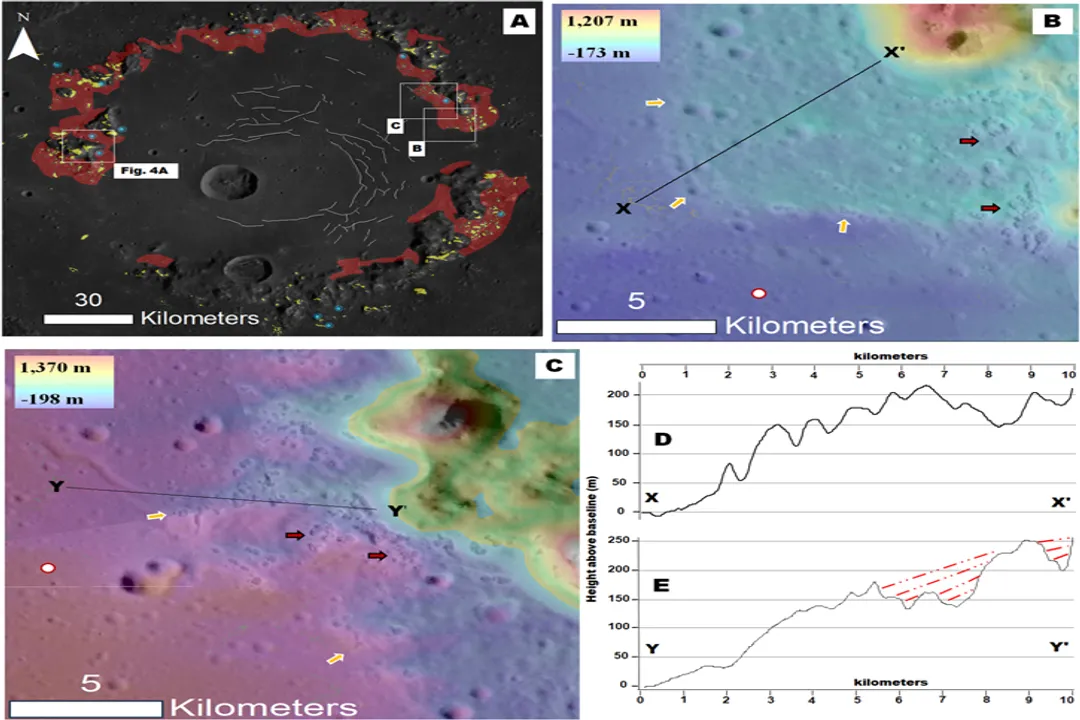
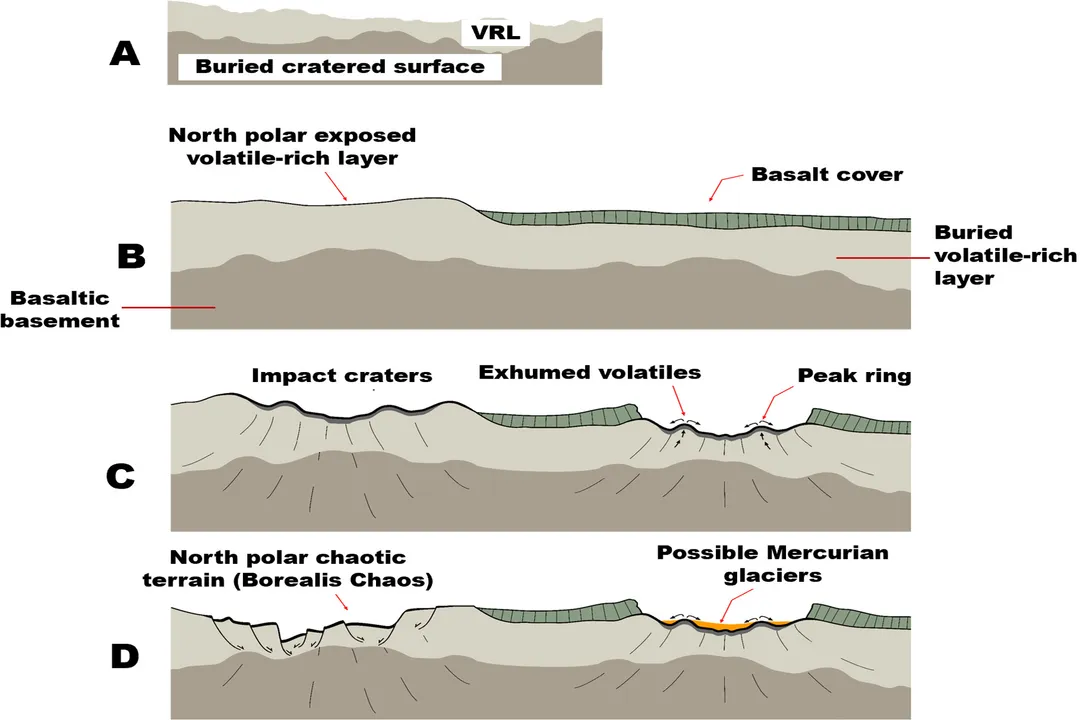
The first detailed compositional data for Mercury came from the Mariner 10 flybys, but it was the MErcury Surface, Space ENvironment, GEochemistry, and Ranging (MESSENGER) mission that revolutionized our understanding by orbiting the planet and mapping its surface chemistry. [1][6][7] MESSENGER's instruments analyzed the surface materials, providing direct evidence about what elements made it to the surface layer. [2]
The data confirmed Mercury is generally rich in silicates, similar to Earth and the Moon, but it exhibits significant differences, particularly in its oxidation state and elemental ratios. [2][8] A key finding has been the detection of elements that are decidedly volatile under solar system formation conditions, suggesting that the planet did not form purely from high-temperature condensates alone. [6]
# Volatiles Detected
The term "volatile elements" covers a wide range, but in the context of planetary science, it often refers to elements with lower condensation temperatures, such as sodium (), potassium (), sulfur (), and zinc (). [2]
# Surface Volatiles
The surface composition revealed by MESSENGER showed that while Mercury is generally depleted in the most volatile elements (like water ice, which is confined to permanently shadowed craters at the poles) compared to Earth, it retains measurable quantities of moderately volatile elements. [1][6]
Potassium () and Sodium () are excellent indicators of surface composition. [2] MESSENGER's X-Ray Spectrometer () detected and across the surface. [1] Interestingly, the ratio of Potassium to Thorium () on Mercury is much lower than what is observed on Earth or the Moon. [2][6][8] Thorium () is a refractory element (it remains solid at high temperatures), so a low ratio implies that Mercury has lost a significant fraction of its original potassium inventory, likely due to high-temperature processing or solar wind stripping over time. [2][6]
Sulfur () is another crucial volatile element detected on Mercury. [2][5] Its presence, particularly in the form of sulfides, suggests that Mercury's formation process involved materials that condensed at lower temperatures than previously assumed for such a close-in orbit, or that the planet's interior allowed these elements to migrate upward to the surface. [5][6] The detection of sulfur, often associated with iron sulfides like pyrrhotite, provides constraints on the redox state (the degree of oxidation) of the material that formed Mercury's mantle and crust. [5]
# Depletion Comparison
To appreciate the significance of the detected volatiles, one must contrast them with the refractory elements. Refractory elements like magnesium (), aluminum (), calcium (), and titanium () are abundant on Mercury's surface. [2][8] This relative enrichment in refractories supports the idea that Mercury formed under very hot conditions, or that subsequent heating events drove off the lighter elements. [8]
Consider the relative depletion: If Mercury had the same bulk composition as Earth, it would be expected to have a much higher abundance of potassium relative to refractory elements. [6] The observed depletion points toward a high-temperature history, possibly one involving a massive impact shortly after formation that melted a large fraction of the planet, allowing the more volatile components to escape into space. [3][7]
# Formation Scenarios

The presence of volatiles alongside a refractory-rich bulk composition forces scientists to consider formation scenarios that can account for this dichotomy. [3][6] One model suggests that Mercury accreted from a high-temperature environment, but perhaps the starting material was already enriched in refractory elements relative to volatiles, or that a massive, high-energy impact stripped away the lighter material later. [1][7]
Another compelling idea involves the Giant Impact Hypothesis or variations thereof, where a massive collision vaporized a significant portion of the proto-planet's mantle, leaving behind a denser, metal-rich core and a silicate shell that was still relatively depleted in the most volatile components. [3]
However, the presence of measurable sodium and potassium, even if depleted relative to Earth, suggests that the high-temperature processing was not total, or that some volatile-bearing materials survived incorporation into the planet itself. [6] The distribution of these elements, which can be mobilized by the solar wind sputtering away surface material, also tells a story about Mercury's long-term interaction with the Sun. [1]
# Surface Dynamics
The surface of Mercury is not static; it is constantly bombarded by the solar wind, which acts as an eroding agent, a process called space weathering. [1] This sputtering effect is particularly important when discussing volatile elements like sodium and potassium. When these elements are ionized on the surface, they can be picked up and carried away by the solar wind, effectively lowering their measured surface abundance over geological time. [1][9]
The MESSENGER mission observed an extended, faint exosphere surrounding Mercury, which is constantly replenished by the surface materials being sputtered away. [1][9] Sodium in this exosphere is particularly bright and detectable, indicating that even though the surface content is lower than expected, there is an active, ongoing loss mechanism at play today. [1]
A point of interest arises when comparing the measured abundance across Mercury's surface to that of other terrestrial bodies. While the Sun's proximity should mandate uniform depletion, the observed variation, though subtle in some contexts, hints at localized processes, perhaps related to ancient volcanism or later, localized resurfacing events, which allowed more volatile-rich material from deeper within the crust to reach the exterior in certain regions. [6] This suggests that the volatile inventory isn't just a relic of the planet's birth; it's also subject to ongoing geological redistribution.
# Volatiles and the Core
The question of volatiles also extends to Mercury's interior, particularly its surprisingly large core. A large core, making up a high percentage of the planet's mass, often implies that a significant amount of lighter, volatile-rich material was lost during formation, leaving behind the denser elements. [7] However, the presence of sulfur in the mantle/crustal silicates raises questions about the sulfur budget of the entire planet. [5] If sulfur was present in the mantle during differentiation, it likely partitioned between the silicate melt and the growing metal core. Analyzing the surface sulfur content helps planetary modelers constrain how much sulfur was locked up in the core versus how much remained in the rocky mantle during the planet's history. [5]
# Insights from Elemental Ratios
The study of elemental ratios is central to distinguishing between different formation theories. Refractory elements provide a baseline for the materials available during condensation. [8]
For instance, the ratio of Titanium () to Aluminum () can indicate how thoroughly the planet melted and differentiated. [2] Since both are refractories, their ratio should be relatively stable across different formation scenarios, unlike the ratio which clearly shows volatile loss. [2][6]
By looking at the abundance of elements like Magnesium () relative to Silicon (), scientists can gauge the degree of vaporization that occurred. [8] A planet that underwent extreme heating and subsequent volatile loss would likely show a different signature compared to one that formed more gently from a cooler reservoir of planetesimals. Mercury's composition suggests a complex history involving significant heating, but not complete vaporization of all components. [8]
If we consider the elemental data in a simplified context, we can imagine a baseline "solar abundance" composition. Mercury’s surface is markedly different from this baseline: it is relatively poor in iron compared to Earth (due to the large core relative to the silicate shell) and, crucially for this discussion, it is significantly depleted in elements that easily boil off, such as potassium. [6][7]
To illustrate the impact of this volatile loss, imagine a thought experiment based on typical solar system ratios. If a hypothetical, pristine Mercury formed with the same ratio as the Sun, and we know the content is roughly consistent with planetary models, the surface we measure today is only a small fraction of that initial budget. [6] This deficit quantifies the amount of volatile material that must have been lost, either during the initial high-temperature accretion or through subsequent processes like massive impacts or intense solar-wind stripping over billions of years. [1][3] The volatile elements that remain are the survivors of this intense chemical editing process.
# Astrobiological Context
While Mercury itself is unlikely to harbor life as we know it due to its extreme temperatures and lack of a substantial atmosphere, the study of its composition has indirect astrobiological implications. [3] Understanding how the most volatile compounds behave under intense stellar irradiation helps define the boundaries of habitability in star systems. [3] If planets forming close to their stars can retain any volatiles—especially those that might have contributed to delivering water or organic precursors to inner worlds like early Earth—it broadens the potential scenarios for planetary evolution elsewhere. [3] The detection of even trace amounts of volatile-bearing materials at the poles, preserved in permanent shadow, suggests that even in the hottest zones of a star system, water ice and other volatiles can persist in shielded niches. [6]
# Element Details
The study of Mercury's chemical makeup often relies on comparing it to more familiar bodies.
| Element Group | Examples | Behavior/Significance | Source Context |
|---|---|---|---|
| Refractory | , , , | Condense at high temperatures; used as reference for bulk composition. | Relatively abundant on the surface. [2][8] |
| Moderately Volatile | , , | Show depletion relative to refractories; indicate high-temperature processing or loss. | Detected on the surface, but is strongly depleted ( ratio). [2][6] |
| Highly Volatile | Water (), (sometimes) | Expected to be almost completely absent from the bulk planet. | Ice is trapped in permanently shadowed regions (PSRs). [6] |
Potassium's apparent depletion is one of the most telling signatures of volatile element loss, as it is a key alkali metal that is easily vaporized compared to the refractory elements that define Mercury's rocky shell. [2][6]
# Surface Weathering Agents
The study of the surface, where we sample these elements, must account for the ongoing weathering process driven by the Sun. [9] The solar wind, a stream of charged particles emanating from the Sun, physically sputters material off the surface, including volatile atoms like sodium. [1][9] This sputtering is so effective that it creates a measurable exosphere that MESSENGER was able to characterize. [1] The composition of this thin, transient atmosphere is dominated by elements that are easily removed from the solid rock, providing a real-time chemical monitor of the planet's interaction with its star. [9]
An interesting deduction stems from considering the interaction between the surface and the solar wind. If we were to take a sample of Mercury's surface rock from a location that has not been recently exposed to intense space weathering—perhaps deep within a large impact crater that shielded the rock for eons—we might expect to find a slightly higher native abundance of moderately volatile elements like potassium than in the general average. [2] This difference highlights that the measured surface composition is a convolution of the planet's ancient history (how it formed) and its present-day interaction with the Sun (how it loses material). [1]
In summary, the volatile elements on Mercury—primarily sulfur, potassium, and sodium—are present as survivors, not as primary components of a low-temperature accretion disk. [2][5] Their observed abundances, when contrasted with refractory elements, paint a picture of a planet that underwent intense thermal processing, causing significant loss of lighter materials, followed by billions of years of solar wind erosion that continues to strip away the remaining volatile signatures from the exposed surface. [1][3][6] The detailed mapping of these surviving elements provides the essential constraints needed to refine models of how terrestrial planets assemble so close to a powerful star. : [8][1] http://ui.adsabs.harvard.edu/abs/2018mvam.book...30N/abstract: [2] https://pubs.geoscienceworld.org/msa/elements/article/15/1/33/568750/The-Surface-Composition-of-Mercury: [3] https://depts.washington.edu/astrobio/wordpress/2023/10/10/the-origin-of-mercurys-structure-and-chemical-composition-and-their-astrobiological-implications/: [4] https://en.wikipedia.org/wiki/Mercury_(element): [5] https://www.sciencedirect.com/science/article/abs/pii/S001910352100350X: [6] https://www.planetary.org/articles/3067: [7] https://www.ncbi.nlm.nih.gov/books/NBK499780/: [8] https://agupubs.onlinelibrary.wiley.com/doi/full/10.1029/2012GL051711: [9] https://iopscience.iop.org/article/10.3847/PSJ/acf219
#Citations
The Chemical Composition of Mercury - NASA ADS
The Surface Composition of Mercury | Elements | GeoScienceWorld
The origin of Mercury's structure and chemical composition and their ...
Mercury (element) - Wikipedia
Volatiles on Mercury: Hollows & Tyagaraja crater vents
Early MESSENGER science results: Mercury is its own planet, not ...
Mercury and Mercury Compounds - NCBI - NIH
Is Mercury a volatile‐rich planet? - McCubbin - 2012 - AGU Journals
Mercury's Hidden Past: Revealing a Volatile-dominated Layer ...