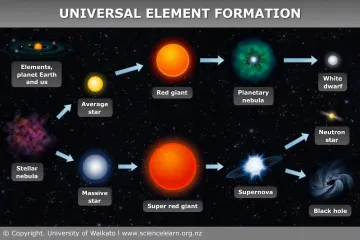
What elements are created by dying stars?

The atoms that constitute our bodies, from the calcium in our bones to the iron in our blood, were not manufactured on Earth. They are ancient artifacts, forged in the extreme thermonuclear laboratories within stars that lived and died long before our solar system even began to coalesce. [8][9] This process, known as stellar nucleosynthesis, describes the creation of atomic nuclei from pre-existing nucleons (protons and neutrons). [2][6] Understanding what elements emerge depends entirely on the life cycle and eventual demise of the star itself.
# Fusion Basics

For most of a star’s life, it exists in a stable, main-sequence phase, sustained by the fusion of hydrogen into helium in its core. [2] This is a relatively low-energy, long-lasting process. Our own Sun, a moderately sized star, is currently performing this essential, foundational step. [5] While this phase creates helium, it doesn't generate the diversity of elements we observe throughout the cosmos. When a star like the Sun exhausts its core hydrogen, it will shed its outer layers and collapse into a dense, hot remnant called a white dwarf. [5] This type of stellar death is quiet in terms of element creation; it does not generate elements heavier than carbon or oxygen through fusion alone. [2]
# Massive Furnaces
The true chemical factories are the much larger, more massive stars, which burn through their fuel supplies at an astonishing rate. [2][4] Once these giants exhaust the hydrogen in their cores, gravity takes over, compressing the core until the temperature is high enough to ignite the next fuel source: helium. [2] This fusion creates carbon and oxygen. [2] In these heavyweight stars, the process doesn't stop there. As the core contracts and heats again, successive layers of fusion begin around the core, creating a stellar onion structure where different elements are fusing simultaneously in shells. [2]
This layered fusion proceeds up the periodic table: carbon fuses to create neon, oxygen, and magnesium; neon fuses to form oxygen and magnesium; oxygen fuses into silicon and sulfur; and finally, silicon fuses to create elements right up to iron. [2][6] Because these reactions occur over millions of years in a relatively controlled (though incredibly hot and dense) environment, the resulting elements—up through iron (atomic number 26)—are produced gradually through stellar nucleosynthesis. [2]
It is insightful to consider the iron limit. Iron is the nuclear endpoint because fusing iron atoms consumes energy rather than releasing it, the very process that keeps a star burning against its own gravity. [2][6] Once the core is pure iron, the star has no more viable fuel source to produce the outward pressure needed to support its immense mass, signaling an imminent catastrophic collapse. [2] This is a stark difference from a smaller star like the Sun, which will cool down after its helium burning phase, never reaching the temperatures required to fuse carbon or higher elements in its core. [5]
# Supernova Ejection
The sudden halt of energy production in a massive star’s iron core triggers an instantaneous collapse, which rebounds in a colossal explosion known as a Type II supernova. [2][8] It is this violent finale that is responsible for creating nearly all the elements heavier than iron. [2][5] The extreme energy and shockwaves generated during the explosion provide the necessary conditions for rapid neutron capture, often called the r-process. [2]
During the r-process, atomic nuclei are bombarded with a flood of free neutrons so quickly that they capture many neutrons before they have time to radioactively decay, resulting in the creation of very heavy, neutron-rich isotopes. [2] Elements like iodine, gold, and uranium are synthesized in these fleeting, catastrophic moments. [2][5][8] This process is distinct from the slower neutron capture (the s-process) that can occur in the later stages of some intermediate-mass stars before they die. [2]
Astronomers have been able to confirm theories about this element forging by directly observing the aftermath of these events. Recently, researchers observed the core of a dying star, capturing direct evidence that confirms the established models of how these heavy atoms are built in the last moments of a star’s life. [7] The energy released in a supernova is immense, allowing the creation of elements that require far more energy input than standard fusion can provide. [4]
# Neutron Star Mergers
While supernovae are responsible for a significant portion of the universe's heavy elements, the very heaviest elements, like gold, platinum, and uranium, may be predominantly synthesized in another type of spectacular event: the collision and merger of two neutron stars. [2]
When two of these incredibly dense stellar remnants, themselves the collapsed cores of previous massive stars, spiral inward and smash together, they generate an environment even more neutron-rich than a supernova. [2] These mergers are thought to be the primary source for the heaviest elements in the cosmos, effectively seeding the universe with the rarest metals we find today. [2]
To contextualize the cosmic supply chain for creating a rocky world like Earth, one can summarize the elemental origins:
- Hydrogen and Helium: Made in the Big Bang. [9]
- Light Elements (up to Carbon/Oxygen): Created during the main-sequence and giant phases of low-to-intermediate mass stars. [2]
- Iron Group Elements: Forged through stable fusion shells in the cores of massive stars before they go supernova. [2]
- Elements Heavier than Iron: Primarily created in the shockwaves of supernovae or the merger events of neutron stars. [2][8]
This cosmic division of labor means that the material available to form a new generation of stars, planets, and eventually, life, is entirely dependent on the diversity of stellar deaths throughout cosmic history. [4]
# Dispersal Into Space
A star’s role is not complete simply by creating these heavy elements; it must also distribute them. [4] The explosive nature of a supernova blasts the newly synthesized materials—the carbon, oxygen, silicon, iron, gold, and everything in between—out into the interstellar medium at incredible speeds. [2][8] The earlier death of a smaller star releases its material more gently, often through a planetary nebula, scattering lighter elements like carbon and nitrogen. [5]
These ejected elements mix with existing gas clouds in space, becoming the raw ingredients for the next generation of stars and planetary systems. [4] When our Sun formed about $4.6$ billion years ago, it incorporated this enriched dust and gas, which is why our solar system contains rocky planets rich in iron and silicates, and why life here can utilize elements heavier than hydrogen and helium. [8] In essence, every time a massive star dies, it enriches the cosmic neighborhood, making the formation of chemically complex bodies possible. [4] The observations made by telescopes like the Spitzer Space Telescope continue to monitor these stellar remnants, helping scientists piece together the timeline and mechanics of this ongoing galactic recycling program. [3] The entire chemistry of our existence is thus tied directly to the final, dramatic moments of stars that lived and died billions of years ago. [8]
#Citations
The Death of a Star - How Stars Work - Science | HowStuffWorks
Stellar nucleosynthesis - Wikipedia
Dying Stars - Spitzer - Caltech
What Did Stars Give Us? - OER Project
Is my body really made up of star stuff? - StarChild - NASA
DOE Explains...Nucleosynthesis - Department of Energy
Astronomers have glimpsed the core of a dying star
We Are Stardust | AMNH
Cosmic ingredients: How the universe forges elements