What is required to burn fuel in rockets?
The fundamental requirement for a rocket to achieve thrust involves a chemical reaction, specifically combustion, which must be carefully managed within the engine structure. [3] For any type of burning to occur, two primary ingredients are essential: the fuel and the oxidizer. [1][4] This is true whether the reaction takes place on Earth, where atmospheric oxygen might be available, or in the near-vacuum of space. [6] Unlike an automobile engine or a common fire, a rocket engine cannot rely on the surrounding environment to supply the necessary component for oxidation. [2]
# Chemical Reaction

At its heart, rocket propulsion relies on rapid, controlled combustion. [3] Combustion is fundamentally a chemical reaction where a substance—the fuel—combines rapidly with an oxidizer, releasing a large amount of energy, usually in the form of heat and expanding gases. [1][3] For a rocket, the goal isn't just to make heat, but to generate a high-velocity exhaust stream directed rearward, which, by Newton's third law, results in forward thrust. [3] The engine itself serves as the structure, often called a combustion chamber, where these elements are mixed, ignited, and allowed to react. [3]
# Space Requirement
The critical difference between how a car engine and a rocket engine operate becomes starkly clear when considering space travel. [2] Jet engines, for instance, work by ingesting atmospheric air, separating the oxygen, and mixing that with their fuel to sustain combustion. [2] In the vacuum of space, there is virtually no ambient oxygen available for this process. [1][2] Therefore, rockets must be completely self-contained regarding their necessary reactants. [6] If a rocket did not carry its own supply of oxidizer, it would cease producing thrust the moment it left the upper reaches of the atmosphere. [2] This necessity—bringing the oxygen supply—is what fundamentally defines the propellants used in rocketry. [8]
# Fuel Oxidizer
The entire mixture used to generate thrust is termed propellant. [5] This propellant is comprised of two distinct functional components: the fuel and the oxidizer. [5][4] The fuel is the substance that gets oxidized—the material providing the chemical energy—while the oxidizer is the chemical agent that allows the fuel to burn in the absence of air. [6]
The mass implications of this requirement are profound. Consider the sheer volume of oxidizer needed to burn a relatively small amount of fuel. For instance, common chemical propellants might involve liquid hydrogen as fuel paired with liquid oxygen as the oxidizer. [8] The oxidizer often constitutes the majority of the total propellant mass needed for a mission. [8] This presents an ongoing engineering challenge: designers must manage the mass of the oxidizer, which is necessary for the reaction to happen, knowing that every kilogram of oxidizer reduces the final mass available for payload or necessary structural material. [5] It is a necessary trade-off; you need the oxidizer to get off the ground, but carrying it limits how much you can lift in the first place.
# Propellant States
Rocket engines are categorized primarily by the physical state of their propellant when they are loaded into the vehicle. [9] The three main classifications are liquid-fueled, solid-fueled, and hybrid systems. [5][9]
# Liquid Systems
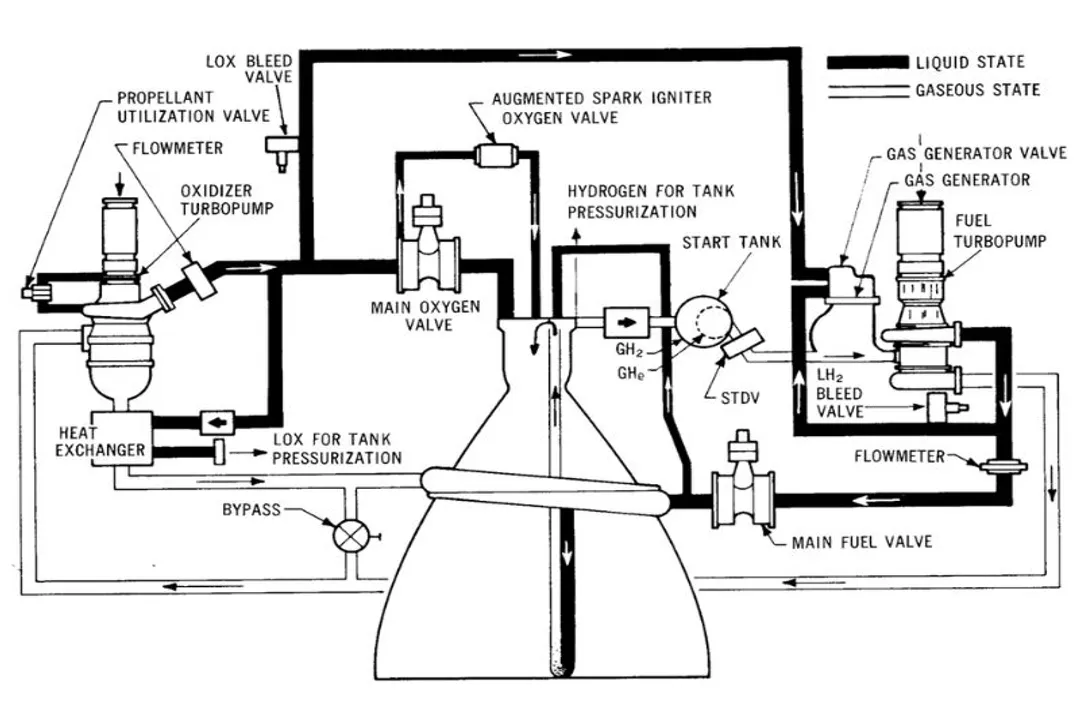
In liquid-fueled rockets, both the fuel and the oxidizer are stored as liquids in separate tanks. [9] Examples often cited include kerosene or liquid hydrogen for fuel, matched with liquid oxygen (LOX) as the oxidizer. [8] These liquids are then pumped at high rates into the combustion chamber where they mix and ignite. [9]
# Solid Systems
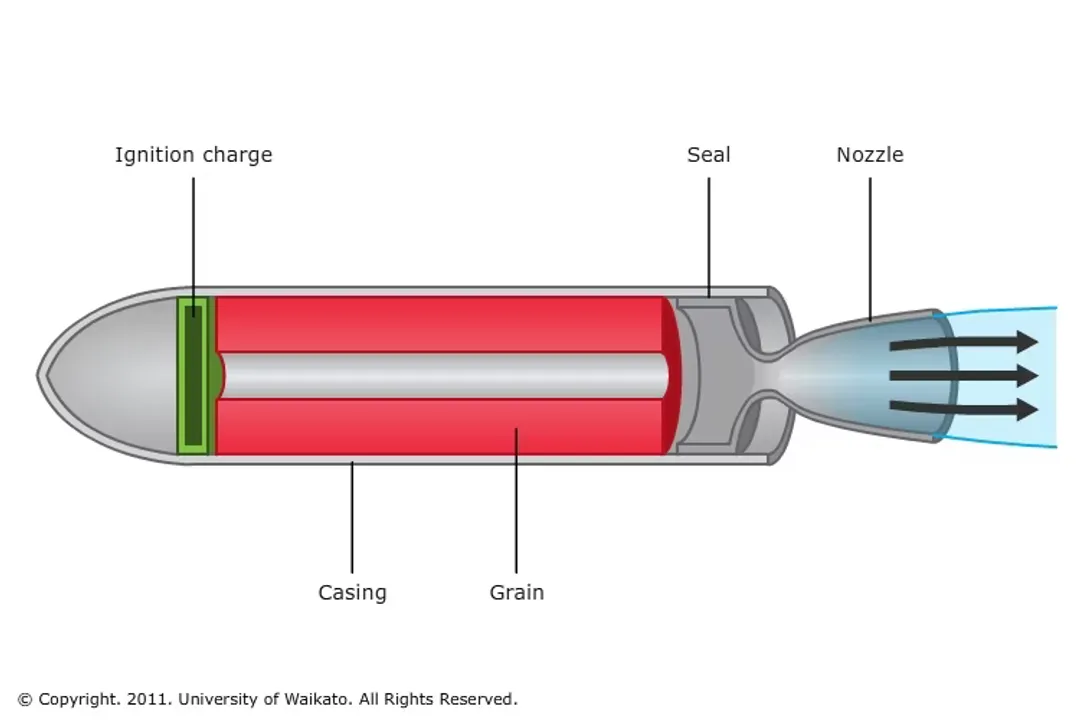
Solid propellant engines are simpler in configuration. Here, the fuel and the oxidizer are already mixed together into a single, rubbery-looking compound that fills the casing of the rocket motor. [9] Once ignited, the entire charge burns from the inside out. [9]
# Hybrid Motors
Hybrid engines represent a compromise, using one component in a liquid or gaseous state and the other in a solid state. [9] For example, a solid fuel might be used with a liquid or gaseous oxidizer pumped over it. [9]
# Burn Control
The physical state of the propellant directly dictates how the engine can be controlled during flight. [9]
Liquid engines offer greater throttling capability and the option for engine restart because the flow of both liquid fuel and liquid oxidizer can be precisely metered and stopped using valves. [9] If the flow is reduced, the thrust decreases; if the flow stops, the engine shuts down. [9] This adaptability is highly valued for complex orbital maneuvers or landing sequences.
In contrast, solid motors, once ignited, are generally committed to burning until all propellant is exhausted. [9] Because the fuel and oxidizer are pre-mixed, shutting them down mid-burn is extremely difficult, if not impossible, through conventional means. [9] This is analogous to lighting a very large firework; the process continues until the fuel is consumed. [9] Therefore, the thrust profile of a solid rocket is largely determined by the shape of the internal grain—the structure of the propellant itself—which dictates the surface area exposed to combustion at any given moment. [9]
Thinking about engine design choices provides a practical example of these trade-offs. A designer choosing a solid motor is prioritizing simplicity, high thrust-to-weight ratio at liftoff, and long-term storability over the ability to adjust thrust once the countdown hits zero. [9] Conversely, selecting a liquid system prioritizes precise thrust control, even at the cost of increased mechanical complexity from the necessary pumps, valves, and plumbing required to manage two separate, highly reactive fluids. [3][9]
#Videos
How Do You Make Rocket Fuels? - YouTube
#Citations
How do rockets burn fuel in space if there isnt oxygen in space?
Ask an Explainer - | How Things Fly
Combustion
What do rockets use as fuel? - Facebook
Scientists Say: Rocket propellants - Science News Explores
Does rocket fuel need oxygen to burn? - Quora
How Do You Make Rocket Fuels? - YouTube
Rocket Physics, Extra Credit: Rocket Fuels
Types of chemical rocket engines - Science Learning Hub

