Is rocket fuel made from water?

The concept of using everyday water as the foundational component for powerful rocket fuel captures the imagination, suggesting easy access and perhaps even planetary sustainability for space travel. While water itself is not the conventional, energetic propellant ignited in a combustion chamber, it holds a fascinating, indirect relationship with some of the most efficient chemical rocket propellants known to rocketry. The key to understanding this connection lies in chemistry and the significant energy required to break water down into its constituent parts.
# Propellant Basics

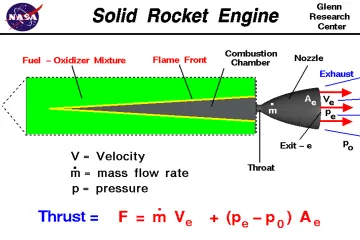
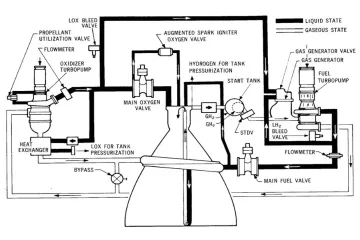
To understand the role of water, one must first clarify what rocket propellant is. A rocket propellant is not simply a single substance that burns; rather, it is a chemical system comprised of two main components: the fuel and the oxidizer. In a vacuum, where there is no external air, the rocket must carry its own source of oxygen to allow the fuel to burn and generate the high-pressure gases needed for thrust. This combination of fuel and oxidizer is stored, mixed, and ignited within the engine's combustion chamber.
Traditional liquid rocket propellants are often categorized by their state—liquid, solid, or hybrid—and their mixture. Common examples include kerosene (like RP-1) mixed with liquid oxygen (), or highly efficient combinations like liquid hydrogen () and liquid oxygen (). These materials possess high specific impulse, a measure of propellant efficiency, because their chemical bonds release a tremendous amount of energy upon reaction.
# Water Split
Water, chemically represented as , is inherently stable, which is precisely why it is not useful as a standalone rocket fuel—it contains virtually no stored chemical energy ready for rapid release. However, water is a compound made of hydrogen and oxygen. If you can separate these two elements, you create the ultimate chemical rocket propellant pair: hydrogen fuel and oxygen oxidizer.
The process of splitting water into its gaseous components is called electrolysis. This reaction involves passing an electric current through water, which breaks the molecular bonds, yielding gaseous hydrogen () and gaseous oxygen (). Since hydrogen and oxygen, when mixed and ignited, react violently to form water again, releasing massive amounts of energy in the process, they are excellent propellants. This combination offers very high performance. Therefore, water serves as a source material for creating one of the best chemical propellants, rather than being the fuel itself.
# Difficulty Energy
The transition from water to rocket-grade propellant is chemically sound but practically daunting because of thermodynamics. Splitting water requires energy input; electrolysis is an energy-intensive, endothermic process. The energy put into the system to separate the hydrogen and oxygen must be less than the energy recovered when those gases are later burned in the rocket engine for the overall system to be worthwhile.
Consider the energy required. To break the strong molecular bonds in water, you need a significant electrical input. Generating this electricity—whether from solar panels, nuclear reactors, or another source—adds mass and complexity to any planned system, especially for in-situ resource utilization (ISRU) on a planet like Mars. The efficiency of this conversion process directly impacts the viability of using water as a propellant source. While the resulting mixture is highly energetic, the energy "cost" of producing it is the primary hurdle for making this a practical, large-scale reality for fueling rockets away from Earth's surface.
It is worth noting that the entire process—electrolysis, liquefaction/storage, and combustion—introduces many points where energy is lost to the environment as waste heat or inefficiencies. If you are planning a mission to, say, the Moon or Mars, and you find frozen water ice, you have a resource, but turning that ice into usable propellant means you must bring the entire power plant with you to run the electrolyzer. For a small-scale demonstration or a future deep-space industrial complex, this might be feasible, but for rapid, high-turnaround launch systems on Earth, transporting the pre-made fuel is currently more energy-efficient overall.
# Steam Rockets
There is a specific, though less common, engine type that does use water directly as a propellant: the steam rocket. These engines operate on a much simpler principle. They do not rely on chemical combustion. Instead, they work by heating water (or superheating it into steam) until it rapidly expands and exits a nozzle, generating thrust.
This heating can be achieved in a few ways. A simple, low-power version uses concentrated solar energy to boil the water, which is known as a solar thermal rocket. A more controllable, but still lower-performance, system uses a chemical heat source, such as a highly concentrated decomposition of hydrogen peroxide (), to superheat the water propellant.
However, the performance—specifically the specific impulse—of steam rockets is considerably lower than that of chemical rockets that burn fuel and oxidizer. While water is abundant and easy to store (as a liquid or solid), the relatively low exhaust velocity means you need a lot more propellant mass to achieve the same change in velocity () compared to using . This limitation generally confines steam rockets to niche applications, such as attitude control or low-thrust maneuvering systems, rather than primary ascent propulsion.
# Propellant Comparison
To truly appreciate the trade-offs, comparing the standard propellant types highlights why the -derived fuel is appealing but difficult.
| Propellant Type | Fuel | Oxidizer | Key Advantage | Key Disadvantage |
|---|---|---|---|---|
| Kerosene/LOX (RP-1) | Kerosene | Liquid Oxygen | High density, storable, reliable ignition | Lower specific impulse than |
| Liquid Hydrogen/LOX () | Liquid Hydrogen | Liquid Oxygen | Highest specific impulse (efficiency) | Extremely low density, requires cryogenic storage ( at ) |
| Water-Derived () | Hydrogen | Oxygen | Source material is abundant in space | Massive energy input required for production |
| Steam Rocket | Water | Water (as reaction mass) | Simple storage, non-toxic | Very low performance (low capability) |
The system is the benchmark for performance, powering the upper stages of many heavy-lift vehicles. When considering water, the initial hurdle is the energy density of the input source required for electrolysis versus the output energy density of the resulting propellant. If a future space-faring civilization has access to abundant, cheap, renewable energy—perhaps vast solar arrays orbiting Mercury or massive fusion reactors—the entire energy economics change. In such a scenario, the ability to "mine" water ice on the Moon or Mars and crack it into fuel in situ becomes incredibly valuable for closing the supply loop and reducing the mass that must be launched from Earth.
# Insight on In-Situ Production
When thinking about the logistics of using water, one might incorrectly assume the entire process must be perfectly energy-neutral or even energy-positive to be worthwhile. However, the real value proposition in space exploration is mass saving. Launching one kilogram of propellant from Earth to orbit might cost thousands of dollars and require a very large rocket to lift the necessary mass of the initial energy source (like a chemical power plant). If, instead, a mission lands on Mars, finds ice, and uses a solar-powered electrolysis unit to generate just one kilogram of , the savings in launch mass from Earth are enormous, even if the electrolysis process itself is only 60% efficient. The net benefit is realized by avoiding the Earth-to-Orbit launch penalty, which is the dominant cost factor in current rocketry. This changes the calculation from "Is it more efficient than Earth-based fuel?" to "Is it more efficient than launching the same mass of fuel from Earth?".
# Rocket Propellant Definition
A final point of clarification involves the definition of propellant itself. The term broadly covers any substance used to generate thrust, whether through chemical reaction or simple thermal expansion. For instance, the United States often defines propellants as materials that do not contain their own oxidizer, meaning they are fuels that require an external oxidizer, such as the liquid oxygen used in many orbital engines. However, propellants can also be monopropellants, which carry their own oxidizer component within the molecule itself, or bipropellants, which are separate fuel and oxidizer components stored distinctly, like the and discussed earlier. Water, being chemically stable and requiring energy input, fits into none of these standard chemical categories directly, reinforcing its status as a resource for fuel rather than the fuel itself.
The dream of rocket fuel from water is therefore partially realized: water provides the atoms ( and ) for an extremely high-performance propellant (), but the process of acquiring those atoms requires a significant, energy-intensive step that is the current roadblock for making it a universally simple or economical choice compared to pre-manufactured chemical systems. The future of space travel may depend heavily on mastering that energy conversion efficiently, especially far from Earth.
#Videos
It's Science! Making rocket fuel - YouTube
#Citations
Making Rocket Fuel From Water - Medium
What is rocket fuel and how do we get it? : r/askscience - Reddit
Rocket propellant - Wikipedia
It's Science! Making rocket fuel - YouTube
How difficult is it to convert water into rocket fuel? - Quora
Scientists Are Getting Closer to a Better Source of Rocket Fuel
What kind of engine uses water as a propellant?
A Quick Guide to Rocket Fuel - Rexarc
Scientists Say: Rocket propellants - Science News Explores