Thermodynamics articles
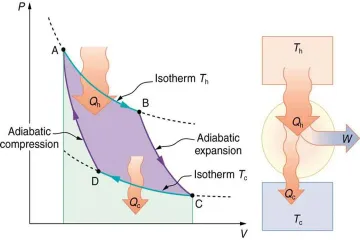
How is the thermal efficiency ($ ext{eta}$) of a heat engine calculated based on energy inputs and outputs?

What fundamental requirement dictates that the efficiency of any real heat engine must always be less than 100%?

What factors exclusively determine the Carnot efficiency, according to the derived formula?

In what temperature scale must $T_H$ and $T_C$ be expressed to correctly calculate Carnot efficiency?

What is the fundamental thermodynamic requirement for extracting work from a heat engine?

Which principle serves as the theoretical foundation proving that no heat engine can be more efficient than a reversible engine operating between the same two temperatures?

Which of the following represents an irreversible process causing real engines to fall short of the Carnot limit?

Besides thermodynamic limits, what practical constraint governs the achievable operating temperature ($T_H$) in modern engines?

How does the Second Law of Thermodynamics differentiate itself from the First Law regarding energy?

If Plant A has $T_H = 900 ext{ K}$ and Plant B has $T_H = 1200 ext{ K}$ (both rejecting at $T_C = 300 ext{ K}$), what does this illustrate about efficiency gains?

What condition must the total entropy of an isolated system meet over time according to the Second Law of Thermodynamics?

Which type of system is defined as one that exchanges energy but prohibits the exchange of mass with its surroundings?

What physical concept is fundamentally provided by the Second Law of Thermodynamics?

If entropy decreases locally in a non-isolated system, what consequence must hold true regarding the total entropy?

According to statistical mechanics, what quantity does entropy ($S$) mathematically relate to via Boltzmann's formula ($S = k \ln \Omega$)?

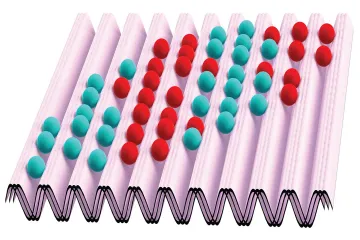
Why does a gas spread out to fill an entire container when a divider is removed, rather than staying clumped on one side?

How does the dispersal of energy (increase in entropy) affect the usefulness of that energy for performing work?

What defines a reversible process in the context of the Second Law?

When considering the Earth-Sun system as an isolated entity, how is the local ordering of life sustained?

What term describes the theoretical final state of the universe if the unidirectional increase in entropy continues indefinitely?
What state does a system naturally seek to minimize at constant temperature and pressure?

Which factor dominates the stable phase determination at very low temperatures?

What characterizes a first-order phase transition concerning energy exchange?

What physical quantities does the Clausius-Clapeyron equation relate on a P-T phase diagram?

What characteristic describes a quantity used as an order parameter in phase transitions?

What event is the ordering upon cooling frequently associated with?

Why must solid-liquid transitions always be separated by a first-order transition line on the phase diagram?

What happens to a thermodynamic response function, like heat capacity, during a second-order phase transition?

What is the real cause for the breakdown of simple models like Mean Field Theory near the critical point (Tc)?
