carbonic acid articles

What is the fundamental characteristic regarding the storage or bottling of carbonic acid?

What two substances react to fundamentally form carbonic acid?

Why does carbonic acid cause a milder effect on $ ext{pH}$ compared to hydrochloric acid?

The familiar fizz sensation in carbonated beverages is directly due to the presence of which substance?

What happens to the concentration of carbonic acid in soda when the bottle is chilled?

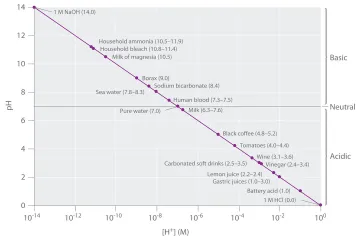
What narrow $ ext{pH}$ range must human blood typically maintain for essential bodily functions?

How does the respiratory system influence blood acidity related to carbonic acid regulation?

Besides manufactured drinks, where is carbonic acid naturally present?

What geological features are formed over vast timescales due to carbonic acid interaction with minerals like calcium carbonate?

What is an advantage of using carbonic acid over strong mineral acids in certain surface preparation tasks?
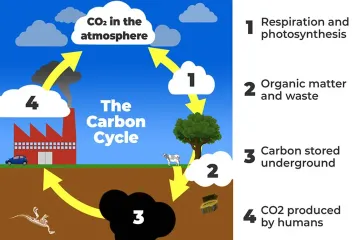
Which reservoir holds the largest approximate amount of carbon according to the provided estimates?

What is the primary natural mechanism in the slow carbon cycle that removes atmospheric $ ext{CO}_2$?

What negative consequence results from the ocean absorbing excess atmospheric $ ext{CO}_2$?

What process is central to the fast carbon cycle, involving the absorption of atmospheric $ ext{CO}_2$ to create sugars?

What is the only major natural pathway for deep, geological carbon stored in rocks to return to the atmosphere?

How does the annual carbon flow rate of the fast biological cycle compare to that of the slow geological cycle?

What is the consequence of thawing permafrost in the far north regarding greenhouse gas emissions?

Approximately how long does it take to lock down carbon absorbed via fast cycles into stable, long-term chemical forms?

Which process, sometimes called carbon fertilization, is leading to enhanced terrestrial uptake of atmospheric $ ext{CO}_2$?

What was the approximate atmospheric $ ext{CO}_2$ concentration before the Industrial Revolution?
At its most basic level, what does an acid-base reaction describe?

What limitation of the Arrhenius theory is highlighted by the behavior of ammonia ($ ext{NH}_3$)?

According to the Brønsted-Lowry theory, how is an acid defined?

In the reaction $\text{CH}_3\text{COOH} + \text{H}_2\text{O} \rightleftharpoons \text{CH}_3\text{COO}^- + \text{H}_3\text{O}^+$, what does the acetate ion ($ ext{CH}_3\text{COO}^-$) represent?

What process involving water allows it to act as both an acid and a base in pure water?

What is the defining molecular event in a Lewis acid-base reaction?

Which species is defined as an electron-pair donor in the Lewis theory?

What is the relationship between Brønsted-Lowry acids and Lewis acids?

When writing the net ionic equation for aqueous neutralization, what are spectator ions?
