How does an inhibitor differ fundamentally from a substrate in an enzyme-catalyzed reaction?
Answer
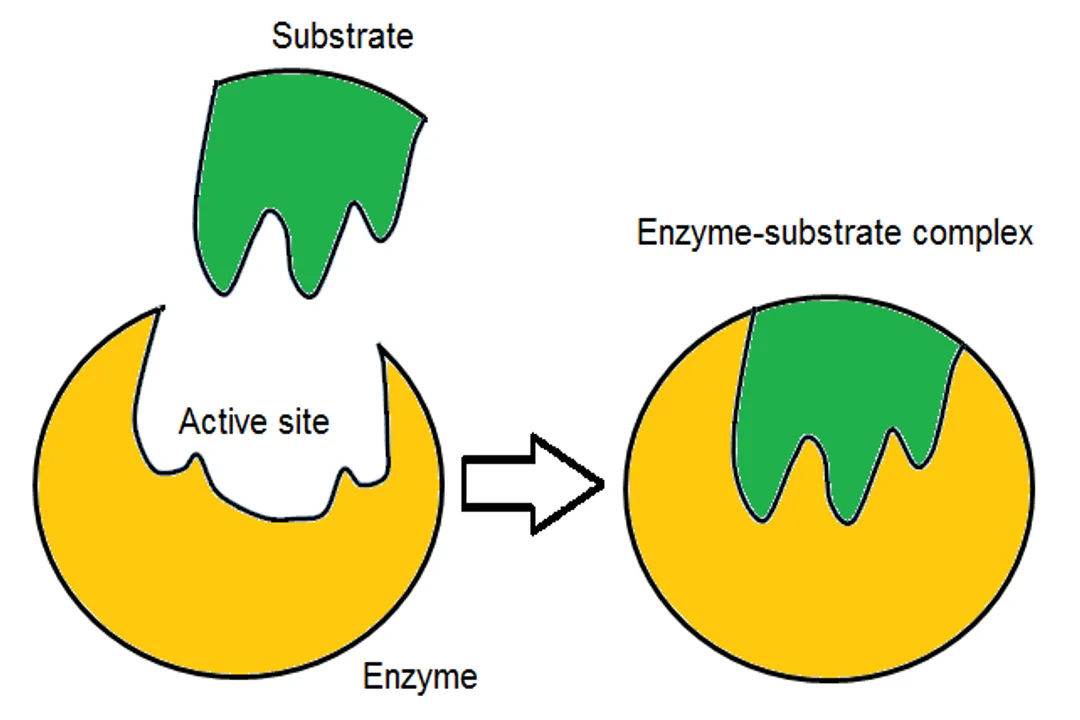
An inhibitor slows or stops the reaction by preventing substrate binding, whereas the substrate is consumed and converted to product.
While a substrate is consumed and converted to a product, an inhibitor binds to the enzyme, often at the active site, preventing the actual substrate from binding and thus slowing or stopping the reaction.
Related Questions
In enzyme-catalyzed reactions, what role does the substrate fulfill?What happens to the reaction rate when substrate concentration becomes very high?What common mechanism is used to describe the relationship between an enzyme and its specific substrate?How does an inhibitor differ fundamentally from a substrate in an enzyme-catalyzed reaction?What material typically serves as the substrate in microelectronics manufacturing?In cell culture, what characteristic of the substrate is crucial for cell adhesion and viability?What is the key difference between a growth medium and a substrate in biological contexts like hydroponics?Generally, what role does the substrate often play in the initial setup of an interaction?If substrate concentration is low, what happens when it is increased?What is described as 'non-negotiable for reproducible results' across both semiconductor preparation and biochemistry?Which property is critical for a microchip substrate but largely irrelevant when an enzyme acts on its substrate?Outside of enzyme action, how is the term substrate commonly used in general non-enzymatic chemical reactions?