What is an orbital in simple terms?
The concept of an atomic orbital often gets tangled up in outdated analogies, primarily because the reality of electron location is governed by the strange rules of quantum mechanics, not the predictable paths of planets orbiting a star. [2][5] Simply put, an orbital is not a defined route or track the electron follows; instead, it is a region of space around the atomic nucleus where the probability of finding an electron is high. [1][3][5] Think of it less like a single path and more like a three-dimensional statistical map showing the electron's most likely whereabouts. [6] In formal terms, these regions are defined by mathematical wave functions derived from solving the Schrödinger equation for the atom. [5][7]
# Probability Zone
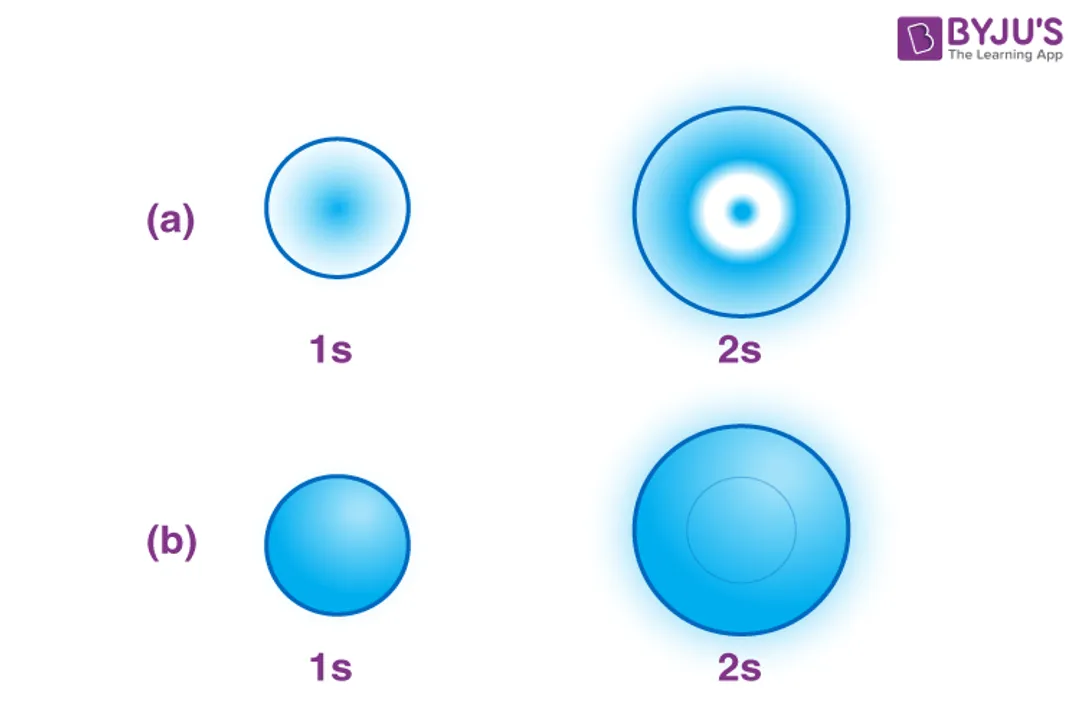
When we discuss where an electron exists, we are dealing entirely in probabilities. [5] Classical physics failed when trying to describe electrons within atoms because an orbiting negative charge should constantly lose energy and spiral into the positive nucleus. Quantum mechanics solved this by defining probability distributions instead of exact locations. [7] An orbital typically encompasses the space where there is a 90% chance of locating the electron. [5] If you could somehow take an extremely long-exposure photograph of an electron in a ground state, the resulting smudge—the cloud—would approximate the shape of the orbital. [2] This differs significantly from the older, familiar model where electrons orbit the nucleus in neat, defined paths, like satellites. [2][6]
# Quantum Rules
The mathematical underpinnings of atomic orbitals are entirely dictated by a set of rules known as quantum numbers. [1] These numbers emerge naturally as solutions to the wave equation describing the electron's behavior within the atom's electric field. [5] A specific orbital is uniquely identified by a unique set of these quantum numbers, acting as its address in the atomic structure. [1] Understanding these numbers is essential to understanding how different orbitals differ from one another in terms of energy, shape, and orientation. [5]
# Principal Number
The first quantum number is the principal quantum number, denoted as . [5] This number dictates the electron's primary energy level or shell. [1][5] The value of must be a positive integer ( and so on). [5] As increases, the orbital becomes larger, and the electrons occupying it are, on average, farther from the nucleus and possess higher energy. [1][10] The shell is the smallest and lowest in energy, holding only one type of orbital. [5]
# Shape Number
The second key number is the angular momentum quantum number, . [5] This number describes the shape of the orbital and defines the subshell. [1][5] Its possible values depend on ; can range from zero up to . [5]
We use letters to designate the shapes associated with the value:
- corresponds to an s-orbital. [1][5]
- corresponds to a p-orbital. [1][5]
- corresponds to a d-orbital. [5]
- corresponds to an f-orbital. [5]
The number of subshells available within any given main energy level () is equal to . [1] For example, when , the possible values are $0, 1,$ and $2$, meaning the third shell contains and subshells. [5]
# Orientation Number
The third number is the magnetic quantum number, . [5] This number specifies the orientation of the orbital in three-dimensional space. [1][5] The possible values for range from through $0$ to . [5] This means that the number of orbitals within a subshell is . [1]
For instance:
- If (-subshell), . There is only one -orbital. [1][5]
- If (-subshell), can be $-1, 0,$ or $+1$. There are three -orbitals. [1][5]
- If (-subshell), can be $-2, -1, 0, +1,$ or $+2$. There are five -orbitals. [5]
This spatial orientation is critical in chemistry, as it determines how atoms approach each other to form bonds. [6]
# Spin Number
The final quantum designation is the spin quantum number, . [5] This number describes the intrinsic angular momentum of the electron, which is often simplified as the direction the electron "spins". [1] It only has two possible values: (often designated as "spin up") or (designated as "spin down"). [5]
# Orbital Shapes
The shapes are perhaps the most visually distinct characteristics of atomic orbitals, directly resulting from the quantum number. [5]
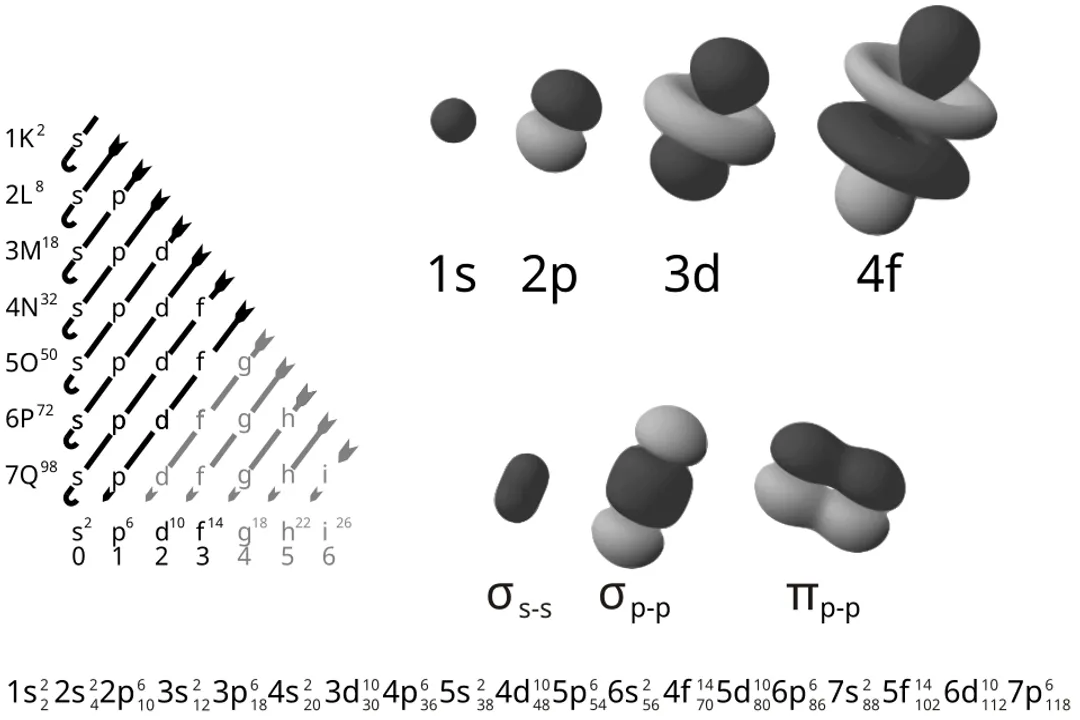
The -orbital () is the simplest: it is perfectly spherical. [1][10] The probability of finding the electron is equal in all directions originating from the nucleus. [10] The orbital is the smallest spherical shape, residing in the first energy level (). [5]
The -orbitals () are characteristically dumbbell-shaped. [1][5][10] They consist of two lobes separated by a plane of zero electron density called a node. [5] Because there are three possible orientations (), there are three -orbitals in every shell starting from (e.g., ). [5][10] All -orbitals within the same energy level have the same energy, but they point along the different Cartesian axes. [1]
The -orbitals () have even more complex shapes, usually resembling four-leaf clovers, with the exception of one that looks like a dumbbell with a donut around the center. [1][5] There are five -orbitals in each subshell, beginning at . [5]
# Filling the Space
The rules governing how electrons occupy these specific three-dimensional spaces are just as important as the shapes themselves. [1] This leads directly to the Pauli Exclusion Principle, which states that no two electrons in an atom can have the exact same set of all four quantum numbers. [1][5] Because the three spatial quantum numbers () define the orbital itself, this principle translates simply to: a single orbital can hold a maximum of two electrons, and those two electrons must have opposite spins ( and ). [1][5]
This electron capacity rule dictates the structure of the periodic table. For example, the shell has one orbital (holding 2 electrons) and three orbitals (holding electrons), for a total capacity of 8 electrons in that shell. [1]
When considering how electrons fill multiple orbitals of the same energy—like the three orbitals—we apply Hund's Rule. This rule dictates that electrons will occupy separate orbitals within a subshell singly, with parallel spins, before pairing up in any one orbital. [5] This minimizes electron-electron repulsion, creating a more stable configuration.
To illustrate the hierarchical structure that results from these rules, consider the size and energy levels:
| Principal Number () | Value(s) | Subshell Letter | Max Orbitals () | Max Electrons |
|---|---|---|---|---|
| 1 | 0 | 1 | 2 | |
| 2 | 0, 1 | $1 + 3$ | 8 | |
| 3 | 0, 1, 2 | $1 + 3 + 5$ | 18 | |
| 4 | 0, 1, 2, 3 | $1 + 3 + 5 + 7$ | 32 | |
| [1][5] |
If you observe the structure of the periodic table, you see this pattern reflected in the block filling: the -block has 2 columns, the -block has 6 columns, and the -block has 10 columns. This direct correspondence shows how fundamental the concept of the orbital shape and occupancy is to predicting chemical behavior. [6]
It is sometimes helpful to visualize the energy difference between these regions. While the and orbitals are in the same principal shell (), the orbital is lower in energy because its probability distribution keeps the electron closer to the nucleus overall, benefiting from stronger attraction compared to the more diffuse orbital. When an atom transitions from an excited state back to the ground state, the electron drops down into the lowest available energy orbital, releasing a photon whose energy matches that specific gap between the old and new orbital energies. [10] This quantifiable energy difference between, say, a orbital and a orbital is what chemists use to predict bond strength and molecular geometry, as the orbitals available for bonding dictate the entire three-dimensional arrangement of atoms in a molecule.
# Beyond the Simple Picture
While and describe the standard atomic orbitals, it is worth noting that in molecules, these atomic orbitals merge and redistribute themselves into molecular orbitals. [7] This process, called hybridization, is necessary because the simple and shapes often cannot explain the observed geometry of molecules like methane (), which has four equal bonds pointing toward the corners of a tetrahedron. To form these, the single carbon atom mathematically mixes its one orbital and its three orbitals to create four new, identical, hybrid orbitals pointing precisely where the hydrogen atoms need to attach. [6] This blending of mathematical solutions is how simple atomic concepts scale up to explain complex chemical architecture.
#Videos
Orbitals, the Basics: Atomic Orbital Tutorial — probability ...
#Citations
Atomic orbital
Simple Explanation of orbitals
Orbitals, the Basics: Atomic Orbital Tutorial — probability ...
Atomic Orbitals and Quantum Numbers
Atomic Orbitals
what is an orbital : r/OrganicChemistry
What is an orbital?
What is orbital? And orbital can accommodate how much ...
ORBITAL Definition & Meaning
Orbitals

