solution articles

What is the typical particle diameter range for dispersed particles in a true solution?

What range defines the particle size for dispersed matter in a colloid?

How is a true solution classified regarding its microscopic uniformity?

Why does a beam of light become visible when passing through a colloid (Tyndall effect)?

What happens to a light beam passing through a true solution?

Which physical phenomenon is primarily responsible for keeping colloidal particles suspended against gravity?

Which specialized technique can be used to separate particles in a colloid but not a true solution?

Which substance is identified in the text as an example of a colloid?

What characteristic defines particles found in a suspension in relation to colloids?

Despite often appearing uniform, how is a colloid technically classified regarding its mixture structure?
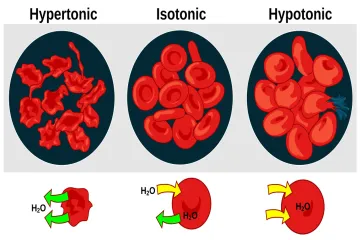
What fundamentally drives the movement of water across a barrier during osmosis?

What specific type of membrane is required for osmotic pressure to manifest?

How is the actual osmotic pressure (Π) defined in relation to external force?

What is identified as the primary driving force behind the flow of the solvent during osmosis?

Which mathematical law serves as an analogy for modeling osmotic pressure in its basic form?

What does the molar concentration (M) variable represent in the basic van't Hoff equation ($ p = MRT$)?

What classification is given to osmotic pressure because its magnitude depends only on the count of particles, not their type?

What term is introduced in the modified van't Hoff equation ($ p = iMRT$) to account for substances that break apart in solution?

For sodium chloride ($ ext{NaCl}$), which dissociates into two ions, what is its theoretical van't Hoff factor ($i$)?
